KLOW Peptide Protocol: Ingredients, Dosing, and Research
Summary
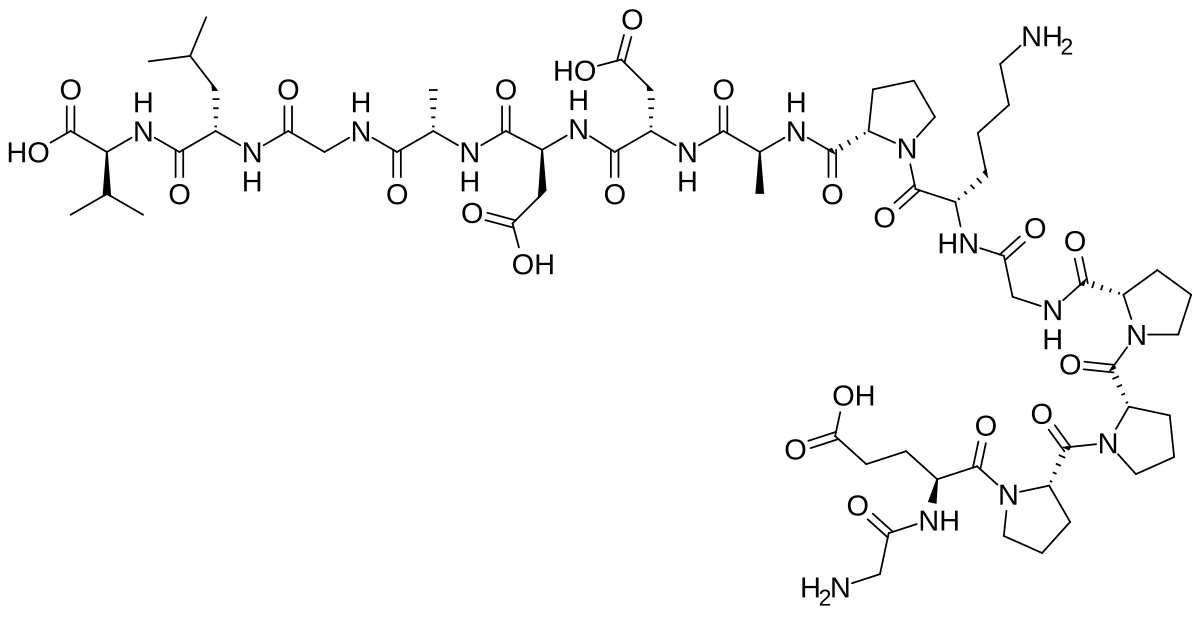
The KLOW peptide protocol is a four-peptide blend combining GHK-Cu (50mg), KPV (10mg), BPC-157 (10mg), and TB-500 (10mg) into a single 80mg vial. It builds on the three-peptide GLOW protocol by adding KPV for enhanced anti-inflammatory action.
What You Need to Know
- Four distinct mechanisms in one injection: collagen synthesis (GHK-Cu), NF-kB inhibition (KPV), growth factor modulation (BPC-157), and cell migration (TB-500)
- No clinical trials exist on the KLOW combination itself. All evidence comes from studies on individual components, none of which are FDA-approved
- Standard dosing: 200-500mcg total blend daily via subcutaneous injection, reconstituted with 3-4mL bacteriostatic water
- Typical protocol length: 4-6 weeks on, 2-4 weeks off. The GHK-Cu component carries a copper accumulation risk with prolonged use
- KLOW vs GLOW: the only difference is the addition of KPV. If chronic inflammation or gut issues aren’t part of your picture, GLOW may be sufficient
Use the blend calculator for KLOW dosing | View full KLOW profile
Four Peptides, Four Mechanisms: Why KLOW Exists
The peptide community didn’t invent KLOW out of thin air. It emerged from the observation that the GLOW protocol (GHK-Cu + BPC-157 + TB-500) worked well for tissue repair and skin rejuvenation but left one gap: it didn’t directly address systemic inflammation.
That’s a problem when healing needs to happen alongside an active inflammatory response. Think autoimmune flare-ups, chronic gut inflammation, or recovery from illness where the immune system is still running hot. BPC-157 and TB-500 both have anti-inflammatory properties, sure, but their primary mechanisms target tissue repair pathways. They reduce inflammation as a secondary effect.
KPV is different. It walks straight into the cell nucleus and shuts down NF-kB, the master switch controlling inflammatory cytokine production (Kannengiesser et al., Anti-Inflammatory Mechanisms, 2020). No other peptide in the GLOW stack does that.
What’s Actually in a KLOW Vial
A standard KLOW vial contains 80mg total across four peptides in a fixed 5:1:1:1 ratio.
| Component | Amount | % of Blend | Role | Research Status |
|---|---|---|---|---|
| GHK-Cu | 50mg | 62.5% | Collagen synthesis, gene modulation | Well-studied individually |
| KPV | 10mg | 12.5% | Anti-inflammatory (NF-kB inhibition) | Emerging research |
| BPC-157 | 10mg | 12.5% | Tissue repair, angiogenesis | Extensively studied (animals) |
| TB-500 | 10mg | 12.5% | Cell migration, wound healing | Well-studied individually |
GHK-Cu dominates the blend at 62.5%. That ratio reflects its naturally lower potency per milligram compared to the other three peptides and the fact that effective GHK-Cu doses (1-3mg daily) are significantly higher than effective BPC-157 doses (250-500mcg daily).
Breaking Down Each Component
GHK-Cu: The Collagen Engine
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is a tripeptide naturally present in human plasma. Your body produces it. Levels decline with age: roughly 200 ng/mL at age 20, dropping to about 80 ng/mL by age 60.
What makes GHK-Cu unusual is scope. A 2018 analysis in the International Journal of Molecular Sciences found it affects 31.2% of human genes, modulating everything from collagen production to antioxidant enzyme expression. A separate 2015 study in Biomedical Research International measured a 70% increase in type I and III collagen and improved elastin production in skin tissue treated with GHK-Cu.
The copper ion isn’t decorative. It’s an essential cofactor for lysyl oxidase, the enzyme that cross-links collagen and elastin fibres. Without copper, those fibres form but don’t properly connect.
Molecular weight: 404.96 Da | Half-life: 0.5-2 hours | Sequence: Gly-His-Lys-Cu2+
KPV: The Inflammation Kill Switch
KPV is a tripeptide derived from the C-terminal fragment of alpha-melanocyte-stimulating hormone (alpha-MSH). It carries alpha-MSH’s anti-inflammatory properties without causing skin darkening. Three amino acids. That’s it.
The mechanism is direct. KPV enters cells and inhibits NF-kB signalling at the nuclear level, reducing production of TNF-alpha and IL-6 (Kannengiesser et al., 2020). A 2021 study on psoriasis models showed 60% reduction in psoriatic inflammatory markers. In IBD models (2019), KPV reduced intestinal inflammation and supported barrier repair.
Critically, KPV doesn’t cause immunosuppression the way corticosteroids do. It modulates inflammation rather than blanket-suppressing immune function. That distinction matters for long-term use.
Half-life: 1-2 hours | Delivery: Injectable, oral, topical, nasal
BPC-157: The Healing Workhorse
BPC-157 has the deepest research base of the four. Over 100 animal studies spanning three decades. It’s a 15-amino-acid synthetic peptide derived from a protein found in human gastric juice, and it works primarily through VEGF-mediated angiogenesis and growth factor expression.
The strength of BPC-157 is breadth. Tendons, muscles, ligaments, gut lining, nerves, blood vessels. A 2017 study demonstrated enhanced VEGF expression and new blood vessel formation at injury sites. A 2020 safety evaluation found no serious adverse effects at 1000mcg/kg for 90 days in animal models.
The weakness is human evidence. Only three published human studies exist as of early 2026, all small pilot studies. A 2024 pilot on interstitial cystitis (12 patients) showed 80-100% symptom resolution. Promising, but far from definitive.
Molecular weight: 1,419.53 Da | Half-life: under 30 minutes | 15 amino acids
TB-500: The Migration Signal
TB-500 is a synthetic fragment of thymosin beta-4, the principal actin-sequestering protein in mammalian cells. It regulates cell migration, which is the process of cells moving toward injury sites.
TB-500 has stronger human data than BPC-157. A 2012 Phase 2 trial in Dermal Wound Healing studied chronic pressure ulcers. A 2016 cardiac trial demonstrated over 50% improvement in left ventricular function post-myocardial infarction. A 2014 Phase 2 trial showed benefit for dry eye (corneal healing).
The longer half-life (approximately 2 hours versus BPC-157’s 30 minutes) means TB-500 distributes systemically. It doesn’t need to be injected near the injury.
Molecular weight: 4,963.44 Da | Half-life: ~2 hours | 43 amino acids
KLOW vs GLOW: Which One?
This is the question everyone asks. The answer depends on what problem you’re trying to address.
| GLOW | KLOW | |
|---|---|---|
| Components | GHK-Cu + BPC-157 + TB-500 | GHK-Cu + KPV + BPC-157 + TB-500 |
| Total vial | 70mg | 80mg |
| Primary focus | Skin rejuvenation, tissue repair | Tissue repair + systemic inflammation |
| KPV included | No | Yes (10mg) |
| Best for | Aesthetic recovery, wound healing, anti-aging | Gut inflammation, autoimmune, complex recovery |
| Injection site reactions | More common (anecdotally) | Less common (KPV may reduce them) |
| Price | Lower (3 peptides) | Higher (4 peptides) |
The practical decision point: is active inflammation part of your picture?
If you’re dealing with a sports injury, post-surgical recovery, or skin rejuvenation without an underlying inflammatory condition, the GLOW protocol covers those bases. Adding KPV to that scenario provides marginal benefit.
If you’re dealing with gut issues (IBD, leaky gut, chronic digestive inflammation), autoimmune flares, or any situation where the immune system is contributing to the problem, the KPV component makes KLOW the more targeted choice. KPV’s ability to inhibit NF-kB at the nuclear level addresses root-cause inflammation in a way the GLOW stack can’t.
Dosing and Reconstitution
Standard Protocol
The KLOW blend calculator handles the maths, but here’s the reference protocol from the community:
Reconstitution: Add 3-4mL bacteriostatic water to the 80mg vial.
- With 4mL: concentration = 20mg/mL total blend
- With 3mL: concentration = 26.7mg/mL total blend
Daily dose: 200-500mcg total blend, subcutaneous injection, once daily.
At 20mg/mL concentration and a 500mcg daily dose, you’re drawing 0.025mL (2.5 units on a standard insulin syringe). That’s tiny. Use a reconstitution calculator if you’re unsure about the volume.
Cycle Structure
- On cycle: 4-6 weeks daily injections
- Off cycle: 2-4 weeks break
- Why cycle: GHK-Cu carries copper accumulation risk with prolonged use. The break allows copper clearance. BPC-157 and TB-500 also lack long-term continuous-use safety data
Reconstitution Steps
- Remove vial cap, swab rubber stopper with alcohol
- Draw 3-4mL bacteriostatic water with a sterile syringe
- Inject slowly down the vial wall — not directly onto the powder
- Gently swirl or roll until fully dissolved. Never shake. Shaking denatures the peptides
- Solution should be clear. GHK-Cu may impart a light blue tint — that’s normal. Green or dark discoloration means contamination
- Label with date and concentration, store at 2-8C (standard refrigerator)
- Use within 4-6 weeks
Open the reconstitution calculator
What the Component Doses Look Like
At 500mcg total KLOW blend daily, the individual component doses break down to:
Those individual doses are below the typical standalone ranges for each peptide (GHK-Cu 1-3mg, KPV 200-500mcg, BPC-157 250-500mcg, TB-500 2-5mg). The theory behind blends is that synergistic effects compensate for lower individual doses. That theory has not been tested in any published study.
Side Effects and Safety Concerns
Reported Side Effects
The KLOW combination hasn’t been studied for safety. What we know comes from individual component research and user reports:
Common (reported frequently in community forums):
- Injection site redness, irritation, or mild swelling
- Light blue-green tint at injection site (from GHK-Cu copper complex — cosmetic only)
- Mild fatigue during the first week
- Temporary digestive changes
Uncommon:
- Headache or light-headedness
- Vivid dreams (commonly attributed to BPC-157)
- Appetite changes
Rare but serious concerns:
- Copper toxicity from prolonged GHK-Cu use — symptoms include metallic taste, fatigue, nausea, abdominal pain. This is why cycling is mandatory, not optional
- Angiogenesis risk: BPC-157 and TB-500 both promote new blood vessel formation. For most people, this is the desired healing mechanism. For anyone with active cancer or a history of cancer, new blood vessel growth could theoretically support tumour development. This is not theoretical for fun — it’s a real pharmacological concern
Contraindications
- Active cancer or history of cancer (angiogenesis risk from BPC-157 and TB-500)
- Wilson’s disease or copper metabolism disorders (GHK-Cu component)
- Pregnancy or breastfeeding (no safety data for any component)
- Active severe infections (theoretical concern with immune modulation from KPV)
- Current anticoagulant therapy (monitor closely — BPC-157 affects blood vessel dynamics)
- WADA-tested athletes: BPC-157 and TB-500 are both prohibited under S0 Unapproved Substances
Expected Timeline
Individual responses vary, but the community-reported timeline looks roughly like this:
| Timeframe | What to Expect |
|---|---|
| Days 1-7 | Subtle inflammation reduction (KPV effect). Some injection site adjustment |
| Week 2-3 | Noticeable decrease in inflammatory symptoms. Early skin texture changes |
| Week 3-4 | Wound healing acceleration, improved gut function (if applicable) |
| Week 4-6 | Full cycle assessment point. Collagen/elastin improvements visible |
| Week 6+ | Diminishing returns begin. Cycle off to clear copper and reset |
The KPV component tends to produce the fastest noticeable effect (days, not weeks) because NF-kB inhibition works quickly. GHK-Cu’s collagen effects take the longest — visible skin improvements often require 6-8 weeks of consistent use.
The Honest Assessment
KLOW is a well-designed blend on paper. Four peptides targeting four distinct mechanisms, combined in a single injection. The individual components all have legitimate research behind them, ranging from extensive (BPC-157’s 100+ animal studies) to emerging (KPV’s handful of preclinical models).
But “well-designed on paper” and “clinically validated” are different things. No one has studied whether these four peptides interact synergistically, antagonistically, or not at all when combined. No one has studied the pharmacokinetics of the blend versus individual administration. No one has established that the fixed 5:1:1:1 ratio is optimal for any specific condition.
That’s not a reason to dismiss KLOW. It’s a reason to be precise about what you know versus what you’re assuming.
If you’re going to run this protocol, use the blend calculator for accurate dosing, cycle properly (4-6 weeks maximum), monitor for copper toxicity signs, and don’t combine KLOW with additional standalone doses of its components. Start conservative at 200mcg/day and titrate up only if needed.
View full KLOW protocol data | Compare with GLOW protocol | Reconstitution calculator
Frequently Asked Questions
What is the KLOW peptide protocol?
KLOW is a four-peptide blend containing GHK-Cu (50mg), KPV (10mg), BPC-157 (10mg), and TB-500 (10mg) in a single 80mg vial. It targets tissue regeneration, collagen synthesis, and systemic inflammation through four distinct biological mechanisms. No clinical trials have studied this specific combination.
What is the difference between KLOW and GLOW peptides?
GLOW contains three peptides (GHK-Cu, BPC-157, TB-500) totalling 70mg. KLOW adds KPV as a fourth component, bringing the total to 80mg. KPV inhibits NF-kB inflammatory signalling, making KLOW better suited for conditions involving chronic inflammation, gut issues, or autoimmune responses. GLOW is sufficient for skin rejuvenation and standard tissue repair.
How do you dose the KLOW peptide blend?
Reconstitute the 80mg vial with 3-4mL bacteriostatic water. Inject 200-500mcg total blend subcutaneously once daily. At 4mL reconstitution volume, a 500mcg dose equals 0.025mL (2.5 units on an insulin syringe). Use a peptide reconstitution calculator for precise volumes.
Is the KLOW peptide protocol safe?
No clinical safety studies exist for the four-peptide KLOW combination. Individual components have varying safety profiles: BPC-157 shows no toxicity in 90-day animal studies at high doses; GHK-Cu carries copper accumulation risk with prolonged use. Both BPC-157 and TB-500 promote angiogenesis, which contraindicates use in anyone with active cancer. Cycle 4-6 weeks on, 2-4 weeks off.
How long should you run a KLOW cycle?
Most protocols recommend 4-6 weeks on, followed by 2-4 weeks off. The mandatory break is primarily driven by the GHK-Cu component — copper accumulates with continuous use and needs clearance time. Some users report noticeable anti-inflammatory effects (from KPV) within the first week, while collagen-related benefits (from GHK-Cu) typically require 6-8 weeks.