BPC-157 vs TB-500: Mechanisms, Dosing & the Wolverine Stack
Summary
BPC-157 and TB-500 are the two most commonly recommended healing peptides. They’re often treated as interchangeable online. They shouldn’t be.
Key Differences
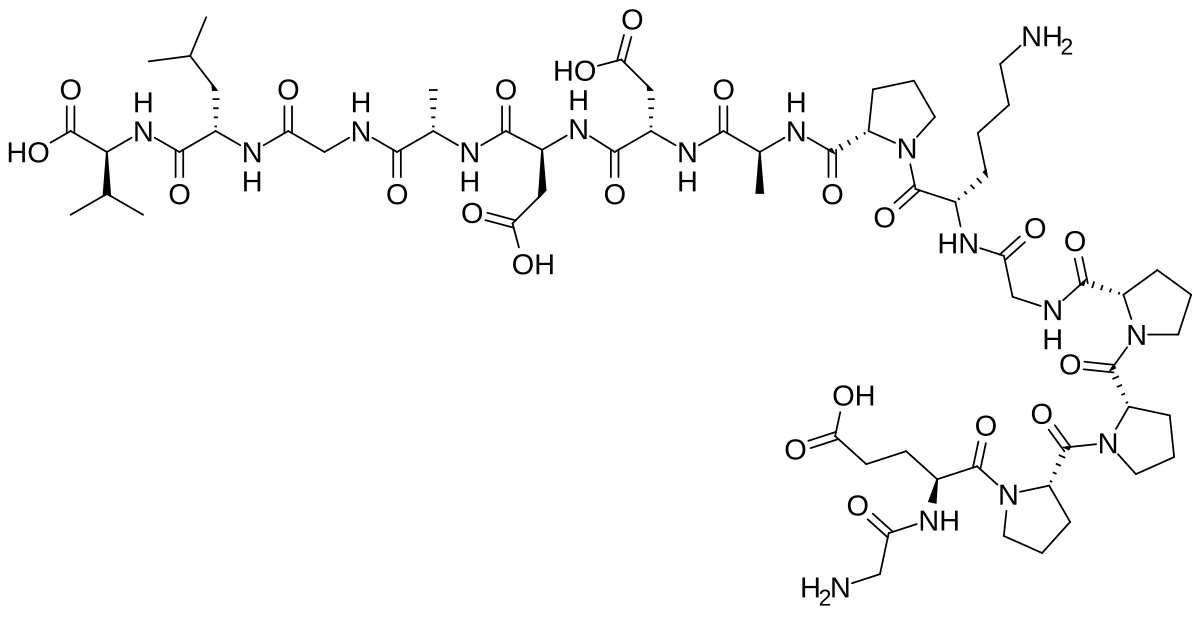
- BPC-157 (1,419 Da, 15 amino acids) drives localized repair through VEGF upregulation and nitric oxide modulation. Dosed at 250-500mcg, once or twice daily. Half-life under 30 minutes
- TB-500 (4,963 Da, 43 amino acids) works systemically by sequestering G-actin to enable cell migration toward injury sites. Dosed at 2-5mg, two or three times weekly. Half-life around 2 hours
- The Wolverine Stack combines both: BPC-157 builds vascular infrastructure at the injury site while TB-500 mobilizes repair cells from across the body
- Neither is FDA-approved. BPC-157 has 3 small pilot studies with fewer than 30 total subjects. TB-500 (as thymosin beta-4) has Phase I, II, and III clinical trial data
When to Pick Which
- BPC-157 first: gut healing, localized tendon/joint injuries, injuries you can inject near
- TB-500 first: systemic muscle damage, post-surgical recovery, multi-site inflammation
- Both (Wolverine Stack): serious tissue damage, healing plateaus, comprehensive recovery
Reconstitution calculator | Accumulation plotter
Different Peptides, Different Jobs
BPC-157 clears your system in under 30 minutes. TB-500 sticks around for about 2 hours. One is dosed in micrograms. The other in milligrams. Molecular weight: 1,419 Daltons versus 4,963. Despite this, peptide forums treat them as interchangeable. They aren’t.
Understanding how each peptide works at a cellular level determines whether you pick one, the other, or stack both. The mechanisms behind the shared “healing peptide” label have almost nothing in common.
How BPC-157 Drives Localized Repair
BPC-157 (Body Protection Compound-157) is a synthetic pentadecapeptide originally isolated from a protective protein in human gastric juice. Sequence: Gly-Lys-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val. Molecular weight: 1,419.53 Daltons. Half-life: under 30 minutes subcutaneously.
That short half-life isn’t a weakness. It reflects the peptide’s role as a localized signaling molecule rather than a systemic drug.
BPC-157’s primary healing pathway runs through VEGFR2. Hsieh et al. (Journal of Molecular Medicine, 2017) demonstrated that BPC-157 activates VEGFR2, which triggers downstream Akt phosphorylation and eNOS expression. The result: new blood vessel formation at the injury site. This is why injecting near the injury matters. You’re directing angiogenesis exactly where tissue needs it.
But blood vessels aren’t the whole picture. A 2025 systematic review by Vasireddi et al. (HSS Journal, 36 studies from 1993-2024) confirmed BPC-157 also upregulates growth hormone receptor expression and reduces inflammatory cytokines in musculoskeletal injury models. Park et al. (Current Pharmaceutical Design, 2020) showed it stabilizes intestinal mucosal barriers and rescues NSAID-induced cytotoxicity. Staresinic et al. (Journal of Orthopaedic Research, 2003) documented accelerated Achilles tendon healing in rats with improved load-to-failure and Young’s modulus values.
The common thread: BPC-157 builds infrastructure at the injury site. New blood vessels, collagen matrix, growth factor receptors. It prepares the ground so the body’s own repair systems can work faster.
BPC-157 human evidence
The human data is thin. Three pilot studies from a single research group in Florida, totaling fewer than 30 subjects.
Lee & Padgett (Alternative Therapies in Health and Medicine, 2021) injected BPC-157 into the knees of 16 patients with chronic pain. 87.5% reported significant relief at 6-12 months. No placebo control. A 2024 interstitial cystitis pilot by Lee et al. treated 12 women with bladder injections and saw 80-100% symptom resolution. A 2025 IV safety study confirmed no adverse effects at doses up to 20mg in two adults.
Small numbers. No randomization or blinding. But the results got attention. For the complete breakdown: BPC-157 Human Clinical Trials.
How TB-500 Works Systemically
TB-500 is a synthetic form of thymosin beta-4, the body’s primary actin-sequestering protein. Molecular weight: 4,963.44 Daltons. Length: 43 amino acids. Half-life: approximately 2 hours. More than three times the size of BPC-157, with a pharmacokinetic window roughly four times longer.
Where BPC-157 builds infrastructure at one location, TB-500 controls traffic across the entire body.
The mechanism centers on G-actin. When tissue is damaged, repair cells need to physically travel to the injury site. That migration requires rapid cytoskeletal remodeling: cells extending pseudopods, reorganizing internal scaffolding, pulling themselves forward through tissue. All of that demands G-actin monomers. TB-500 regulates the supply by sequestering actin into a pool of ready-to-use building blocks.
Goldstein et al. (Expert Opinion on Biological Therapy, 2012) laid out five distinct effects from this one mechanism: cell migration, stem cell mobilization, new blood vessel formation, inflammation reduction, and decreased scar formation. One protein controlling actin availability produces all five downstream results.
TB-500 human evidence
The clinical picture is substantially stronger than BPC-157.
Wang et al. (Journal of Cellular and Molecular Medicine, 2021) published a Phase I dose-escalation study in 54 healthy subjects. Ascending IV doses from 0.05 to 25.0 mcg/kg produced only mild to moderate adverse events. Zero dose-limiting toxicities. Zero serious adverse events.
Treadwell et al. (Annals of the New York Academy of Sciences, 2012) reported two Phase II clinical trials where thymosin beta-4 accelerated healing of chronic pressure ulcers and venous stasis ulcers by approximately one month compared to placebo. Placebo-controlled data. Something BPC-157 doesn’t have.
Sosne et al. (Cornea, 2015) ran a Phase II randomized trial in 9 severe dry eye patients. RGN-259 (0.1% thymosin beta-4 eye drops) achieved statistically significant improvements in both signs and symptoms, with effects persisting 28 days after treatment ended. That same group reached Phase III (International Journal of Molecular Sciences, 2022) with a randomized, double-masked trial demonstrating corneal healing in neurotrophic keratopathy patients.
Phase I, Phase II, Phase III. That’s a clinical evidence trajectory BPC-157 hasn’t approached.
BPC-157 vs TB-500: Head to Head
| Property | BPC-157 | TB-500 |
|---|---|---|
| Molecular weight | 1,419.53 Da | 4,963.44 Da |
| Amino acids | 15 | 43 |
| Source protein | Human gastric juice | Thymosin beta-4 |
| Half-life | Under 30 minutes | ~2 hours |
| Typical dose | 250-500mcg | 2-5mg |
| Dosing frequency | 1-2x daily | 2-3x weekly |
| Primary mechanism | VEGFR2/angiogenesis, NO modulation | Actin sequestration, cell migration |
| Best application | Localized injury, gut healing | Systemic repair, muscle regeneration |
| Human trials | 3 pilot studies (under 30 subjects) | Phase I, II, III (100+ subjects) |
| FDA status | Category 2 (restricted) | Not scheduled |
| WADA status | Prohibited (S0) | Prohibited |
The pharmacokinetic difference explains why these peptides are dosed so differently. BPC-157 peaks fast and clears fast. TB-500 peaks later and lingers.
When BPC-157 is the better choice
BPC-157 excels when the problem is in one place. A torn Achilles tendon, a rotator cuff you can inject near, gastric ulcers, NSAID-damaged gut lining, inflammatory bowel symptoms. Its angiogenic signaling concentrates at the injection site, building blood vessels and collagen matrix where you direct it.
Gastric stability is another advantage. BPC-157 remains active in stomach acid, making oral dosing viable for GI conditions. TB-500 doesn’t survive oral administration reliably.
When TB-500 is the better choice
TB-500 makes more sense for widespread damage. Multi-site injuries, full-body recovery after surgery, chronic inflammation across several joints, athletic recovery where the entire musculoskeletal system is under stress. Its longer half-life and systemic cell-mobilization mechanism cover territory that BPC-157’s localized approach simply can’t reach.
TB-500’s wound healing data is also stronger in absolute terms. Placebo-controlled Phase II trials showing roughly one month of accelerated chronic ulcer healing. BPC-157 has nothing equivalent.
The Wolverine Stack: Running Both Together
The name comes from the Marvel character’s regenerative abilities. It stuck because the BPC-157 and TB-500 combination addresses tissue repair from two complementary angles that don’t overlap.
The biological logic: BPC-157 upregulates VEGF and builds vascular infrastructure at injury sites. TB-500 sequesters G-actin to mobilize repair cells toward those sites. One peptide builds the roads. The other sends the repair crew down them.
Standard protocol
BPC-157: 250mcg subcutaneous, twice daily (morning and evening). Inject near the injury site when accessible.
TB-500: 2-2.5mg subcutaneous, twice weekly (e.g., Monday and Thursday).
Cycle length: 6-8 weeks for active injury recovery.
Optional loading phase: TB-500 at 5mg twice weekly for the first 2 weeks, then reduce to 2-2.5mg for the remaining cycle.
Each peptide is reconstituted separately with bacteriostatic water. Don’t mix them in the same syringe unless using a pre-made blend. Use separate injection sites when dosing on the same day. Calculate your reconstitution volumes here.
Why the dosing looks so different
The asymmetry confuses people at first. BPC-157 in micrograms daily. TB-500 in milligrams weekly. The half-lives explain it.
BPC-157 clears in under 30 minutes. Frequent small doses maintain consistent signaling at the injury site throughout the day. Think of it as a daily signal flare telling the body where to send resources.
TB-500 lasts roughly 2 hours and distributes systemically. Less frequent, larger doses keep adequate tissue-level concentrations through the week. Two or three injections sustain the cell-migration effect without daily dosing.
What about pre-mixed blends?
BPC-157/TB-500 blends exist, including the KLOW protocol which adds GHK-Cu and KPV to the stack. These trade dosing flexibility for convenience. If your injury responds better to higher TB-500, you can’t adjust one peptide without changing the other in a blend.
For most use cases, separate vials provide more control over individual dosing. The reconstitution calculator handles the math for both.
View the full Wolverine Stack protocol data
The Evidence Gap: What’s Real and What’s Extrapolated
The most common mistake in peptide forums is treating preclinical evidence as equivalent to clinical evidence. BPC-157’s 100+ animal studies demonstrate biological mechanism and therapeutic potential in rodent models. They don’t demonstrate safety or efficacy in humans.
Mice heal differently than people. Dose extrapolations from 10 mcg/kg in a rat don’t reliably predict human pharmacokinetics. The only published BPC-157 pharmacokinetic study (Xu et al., Frontiers in Pharmacology, 2022) showed 14-19% intramuscular bioavailability in rats and 45-51% in dogs. No human PK data has been published. The doses people use today are educated guesses based on animal extrapolation and community trial-and-error.
TB-500 sits on firmer ground clinically. But it’s still not FDA-approved for healing or performance applications. The Phase III trial (Sosne et al., 2022) addressed corneal healing specifically, not the musculoskeletal uses most people want it for.
Both peptides need more human data. That’s the honest answer. The question is how much uncertainty you’re comfortable with, and whether the existing evidence justifies the risk for your specific situation.
Safety: Shared Risks and Contraindications
Both BPC-157 and TB-500 promote angiogenesis. That’s their healing mechanism. It’s also the reason active cancer is a hard contraindication for both.
Other shared contraindications include pregnancy, breastfeeding, and concurrent anticoagulant therapy. Both peptides affect vascular growth in ways that may interact with blood thinners.
Reported side effects for both tend to be mild. BPC-157 users note headaches and light-headedness more frequently. TB-500 users report temporary fatigue and occasional lethargy in the first week. Injection site redness and swelling are the most common complaint for both. Neither peptide has produced serious adverse events in published human data.
For a deep dive into BPC-157’s risk profile: BPC-157 Side Effects: Is It Safe?
Both are prohibited by WADA. Competitive athletes should avoid use entirely. The legal status of BPC-157 is also in flux. It was placed on the FDA Category 2 list in 2023, but the 2026 reclassification process may restore compounding pharmacy access. TB-500 (thymosin beta-4) is not on the Category 2 list and has active Investigational New Drug applications.
Frequently Asked Questions
Can I take BPC-157 and TB-500 at the same time?
Yes. The Wolverine Stack runs both concurrently: BPC-157 at 250mcg twice daily alongside TB-500 at 2-2.5mg twice weekly. They work through different receptors and pathways. No negative interactions have been documented. Use separate injection sites when dosing on the same day.
Is TB-500 the same as thymosin beta-4?
Not exactly. Commercial TB-500 may contain the full 43-amino-acid thymosin beta-4 sequence or just the 7-amino-acid active fragment (Ac-LKKTETQ). Most clinical research used full-length thymosin beta-4. The fragment retains the actin-binding domain responsible for cell migration and wound healing but may differ in systemic distribution.
Which one works better for tendon injuries?
BPC-157 has more direct tendon evidence. Staresinic et al. (Journal of Orthopaedic Research, 2003) showed accelerated Achilles tendon healing with improved biomechanical properties in rats. If you can inject near the injured tendon, BPC-157 likely has the advantage. For diffuse connective tissue issues affecting multiple sites, TB-500’s systemic reach covers more ground.
How long should a Wolverine Stack cycle last?
Standard cycles run 6-8 weeks. Inflammation reduction typically starts in week 1-2. Recovery improvements become noticeable around week 2-4. Structural tissue changes peak between week 4-8. Some protocols front-load TB-500 at 5mg twice weekly for the first two weeks before dropping to maintenance doses.
Are BPC-157 and TB-500 legal?
Status varies by country and is changing. BPC-157 sits on the FDA Category 2 list (2023), restricting compounding pharmacy access, though the 2026 reclassification may change this. TB-500 is not on the Category 2 list and has active IND applications for ophthalmic uses. Both are sold as “research chemicals” in a legal grey area. Both are banned by WADA for competitive athletes.